Bioinks & Biomaterials
High-quality bioinks play an important role in the success of 3D cell culture and 3D bioprinting. As the first bioink company, we’re constantly creating and refining bioinks for a variety of application areas. This has allowed countless labs the ability to get started with what really matters: creating the future of health and medicine.
Achieve research success faster with our bioinks.
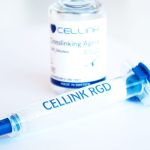
Bioinks
From standard bioinks shipped to tissue-specific formulas shipped in sterile cartridges ready to be used in your research.
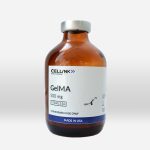
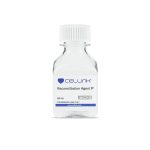
Base materials
With a wide array of stock solutions and lyophilized biomaterials like GelMA, collagen, and alginate, as well as effective thickening and crosslinking agents, we offer unrivaled flexibility for you to develop your own bioinks made from high-quality and consistent building blocks.
Accessories
All the additional accessories needed for you to get up and running with your print.

Nozzles
Choose from a wide range of sterile nozzles, including standard, precision, high precision, and micron precision options, ranging from 18G to 30G
Needles
Explore a diverse selection of sterile needles, available in various gauges, length and multiple precision levels, spanning from 18G to 30G

Cartridges
Sterile cartridges, such as UV-shielding, thermoplastic, and aluminum types, provide efficient protection while maintaining product integrity
For every cell type and every application
To truly capture native cell biology we develop biomaterials that have all the biological cues needed.
Whether it is ECM components that drive stem cell differentiation, or biomechanical properties supporting cardiomyocyte functionality. No matter the cell type, or application our team has a solution.
Bioink Selector Guide
Unsure were to start? Dive into our bioink selector guide to get a recommended starting point for your research.

CARTILAGE CELLS
Bioinks refer to any synthetic or natural polymer selected for its biocompatible components and favorable rheological properties. These characteristics will either temporarily or permanently support living cells to facilitate cell adhesion, cell proliferation and cell differentiation during maturation. Read our full blog by clicking the button below to learn more about bioinks.

Industry-leading, rigorous testing
Commitment to quality
Customer satisfaction and high-quality products
are our highest priorities. We firmly believe that we will only be able to achieve our vision by continuously improving the cost effectiveness and performance of all our products and processes, as well as focusing on innovation. To ensure our customers get the best bioinks, we perform strict assessments of raw material suppliers to ensure materials are endotoxin-free, sterile and consistent from batch to batch.
We produce pure, premium biomaterials that maintain low batch-to-batch variability for reliably reproducible results.
Speak to an expert
As leaders in biomaterial development, benefit from our bioink scientists’ extensive lab experience and profound biomaterial expertise. They are well-equipped to assist you in selecting the perfect bioink to meet all your research requirements.