Bioprinting:
Revolutionizing the search for new cancer treatments and cures
One of the biggest challenges in cancer drug and therapeutics development today is that 85% of drugs in clinical trials fail to reach the market. Clinical trial activity remains a high-risk endeavor as the oncology composite success rate fell to 8% in 2018 from 11.7% in 2017 (Global Oncology Trends 2019, IQVIA).
Bioprinting is revolutionizing the way researchers approach cancer studies by providing more physiologically relevant models for understanding the disease and testing new treatments. With its ability to precisely arrange cells in a three-dimensional environment, bioprinting allows for the creation of multicellular models that closely resemble the native microenvironments found in the human body. Bioprinting is not only helping researchers gain a deeper understanding of tumor formation and progression, but it is also enabling the development of more effective drug discovery studies.
To raise awareness about cancer, a disease that affects so many, in this page we feature key studies conducted by CELLINK customers and scientists, or as we like to call them, Printing Pioneers. Learn how researchers are leveraging bioprinting to advance cancer research and help find new treatments for this devastating disease.
Bioprinting advances brain cancer drug screening

Fabricating glioblastoma-on-a-chip model for high-throughput drug screening and personalized treatments
Dr. Joshua Chou’s group (University of Technology Sydney) is focusing on the understanding glioblastoma, an aggressive brain cancer with a very low survival rate. A challenge compounded by the fact that the complex microenvironment is naturally protected by the blood brain barrier, which limits anti-cancer drugs from diffusing through the barrier.
Currently there are two key problems faced by researchers. The first is the gap where no biomimetic in-vitro models that can be utilized in high-throughput drug screening exist. The second is the lack of models that effectively provide a 3D tumor environment, and capture the complexities created by the blood brain barrier . Utilizing CELLINK’s bioprinting technology, Dr. Giulia Silvani, a member of Dr. Chou’s team, was able to develop a gbm-on-a-chip model that possess all the critical features of the glioblastoma microenvironment and the blood brain barrier. By leveraging the BIO X the group has successfully developed a more realistic glioblastoma model that integrates with high-throughput drug screening workflows.
”Our gbm-on-a-chip model will allow researchers the ability to better understand how drugs diffuse through the blood brain barrier. It will also help to optimize and refine drug formulation to reach effective therapeutic concentration to target the brain cancer. Furthermore, our model provides a platform for future personalized drug screening for brain cancer patients. We can culture patient specific brain cancer cells, recreate them in our model and conduct drug screening to determine, which drug is more effective towards treating the individuals’ brain cancer.”
– Dr. Joshua Chou, University of Technology Sydney
Transforming breast cancer treatment

Patient-specific tumor models for personalized medicine testing
Carcinotech’s advanced 3D-printed micro-tumors provide a platform for rapid, ethical, and accurate drug screening, for pre-clinical and personalized medicine testing. Using patient-derived biopsies, primary cells, immune cells, and cancer stem cells the team has developed 3D-bioprinted micro-tumors for breast cancer that are representative of the tumor microenvironment and the patient’s immune cells.
Carcinotech has developed and expanded their technology into six cancer types including breast, lung, brain, colorectal cancer and more. By using 3D bioprinting the team produces hundreds of breast cancer micro-tumors from a single patient biopsy. This allows for multiple drugs to be tested simultaneously. Combining automation and bioprinting, Carcinotech can accelerate the development of effective treatments for cancer, offering surgeons and oncologists the opportunity to test treatment options and create personalized treatment plans for each individual patient.
”Our living tumours can be used to advance research into new drugs and treatments to aid in the fight against breast cancer, improving the quality of life and chance of survival for those suffering from this disease. We hope that in the future our models will allow every individual the opportunity to receive personal cancer care, improving treatments and increasing their chance of survival.”
– Ishani Malhotra, Founder, Carcinotech
Bioprinting advances colorectal cancer research
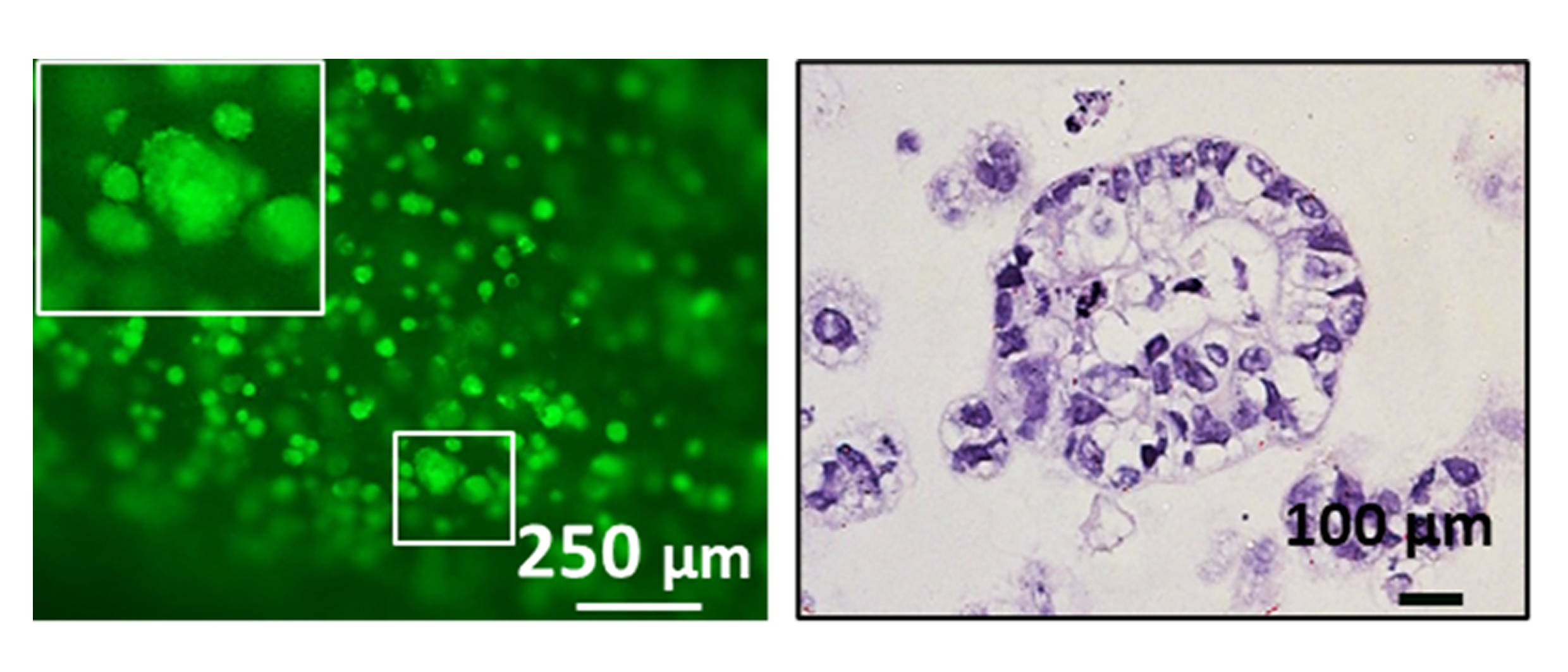
As few as 0.1% of all drugs make it from bench to bedside, significantly impacted by the poor predictive models available during pre-clinical and clinical phases of drug testing. Therefore, the success of developing novel treatment agents lies in the introduction of improved preclinical disease models which resemble in vivo carcinomas closer, possess higher predictive properties, and offer opportunities for individualized therapies.
Dr. Sbirkov and his colleagues from the Medical University of Plovdiv, Bulgaria, have developed a highly reproducible, affordable, and flexible model of colorectal cancer. Bioprinting allowed the researchers to place the cells in 3D structures that would provide the necessary spatial organization and extracellular components to recapitulate the in vivo tumor microenvironment and biology of the cells.
“Having in vitro models, which are more representative to what happens with the cells in the body, would generate more physiologically relevant pre-clinical data with higher predictive power in terms of drug response so ultimately this will improve drug discovery and personalized medicine. “
– Yordan Sbirkov, Medical University-Plovdiv
Advancing Lung Cancer Research
Creating a perfusable 3D model with DLP Bioprinting
Lung cancer continues to pose a major global health challenge, and traditional research models, like 2D cultures and animal studies, fail to capture the complexity of human tumors, contributing to drug failure rates as high as 97% in clinical trials. To address this, Prof. Jens Kurreck and his team at Technische Universität Berlin turned to bioprinting to develop a more physiologically relevant human lung cancer model that can better support the discovery and testing of more effective therapies.
“There is a huge difference in causes of diseases and their underlying mechanisms between animal models and humans. This is why we believe it is important to have human organ models and bioprinting enables us to produce these models with high accuracy.”
– Prof. Jens Kurreck, Technische Universität Berlin
Unlocking the secrets of lung cancer
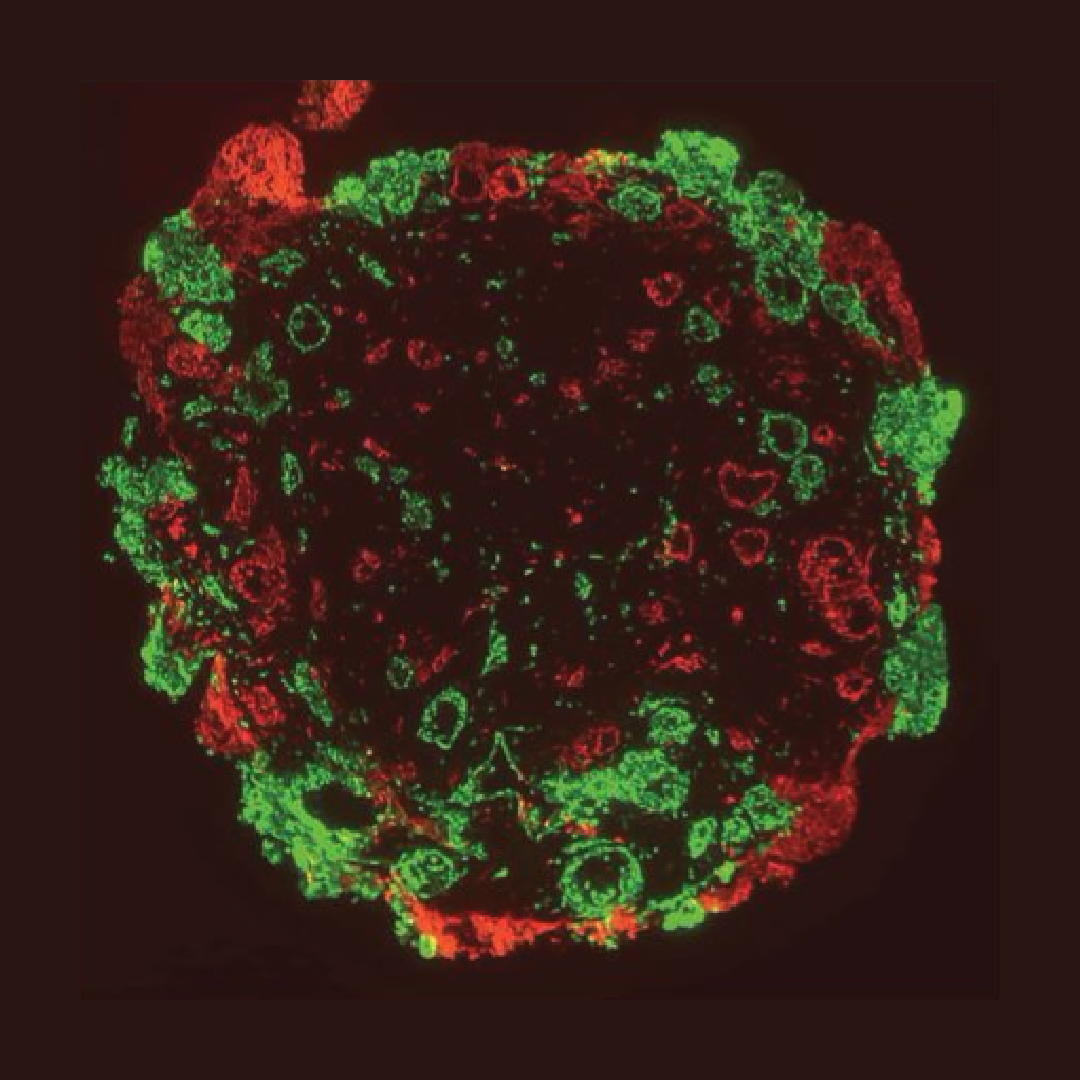
Lung cancer is one of the leading causes of cancer-related deaths in the world. Conventional 3D models, including organoids and spheroids, unfortunately have a major limitation. Their lack of a native microenvionment increases the challenges in capturing cell-cell communication and migration patterns within ECM based stroma. Establishment of such native tumor microenvironment is critical to understand disease progression and developing robust drug discovery studies. To address this challenge a team of researchers at CELLINK fabricated multicellular lung cancer assembloid models.
The team incorporated human lung cancer cells (A549), lung adenocarcinoma-associated fibroblasts, and human umbilical vein endothelial cells in a laminin and collagen rich stromal environment. As a result, they were able to visualize the migration of cancer cells and merging of cancer spheroids within the extracellular matrix (ECM).
Using 3D bioprinting offered multiple advantages, including the precise geometrical arrangement of multicellular constructs that can better recapitulate the native 3D human physiology. Cells self-assemble based on external signals from surrounding cells and the environment. This important phenomenon is aiding in the understanding of tumor formation and may also potentially facilitate drug development and screening.

BIO X™






