How 3D printed lung tissue is transforming lung therapeutics
Kras-driven Lung Cancer by Eric Snyder, 2015
Lung diseases have a high prevalence among the global population. The World Health Organization lists chronic obstructive pulmonary disease (COPD) as the third largest cause of death worldwide, causing 3.23 million deaths in 2019. Not only can lung diseases (such as COPD) cause death, they also contribute to a higher risk of other health problems like cardiovascular disease and infections.
Bioprinting is transforming lung research by enabling researchers to 3D print lung tissue models with greater biomimetic relevance. This method holds great potential for therapeutic applications, disease modeling and tissue regeneration. With the integration of living lung cells into three-dimensional constructs, researchers can recreate the conditions present in lung disease. This enables not only a greater understanding of disease mechanisms, but also the ability to discover more effective treatments, in terms of treatment plans, tissue regeneration and drug efficiency.
In this blog, we highlight how scientists leverage 3D bioprinting to advance lung disease research and tissue regeneration. Explore these breakthroughs and the development of innovative 3D models aimed at combating pulmonary conditions.
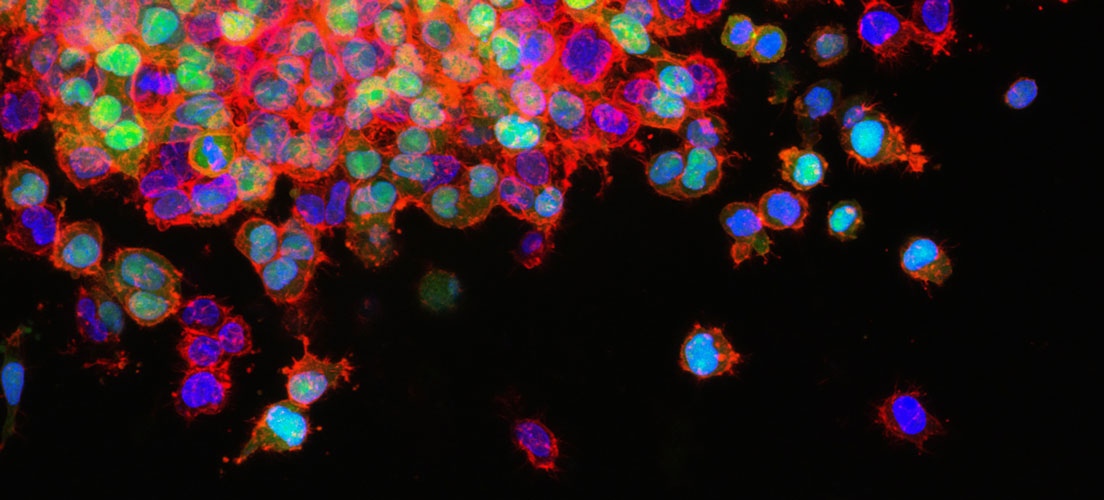
Lung cancer drug resistance testing
Lung cancer-on-chip model for long-term culturing and drug resistance testing
Lung carcinoma is a very aggressive cancer with a very low survival rate. This is in part due to the cancer’s high drug resistance, which often results in its reappearance. One challenge when studying long-term effects of therapeutics and the resistance mechanisms of the cancer is the culturing lifespan.
Dr. Agnieszka Krakos’ research team at Wroclaw University of Science and Technology is seeking to extend the culture lifespan of the H69AR lung cancer cell line, which has a multi-drug resistant phenotype. By extending the cell culture lifespan of this cell line, it will become easier to identify its resistance mechanisms, and to evaluate current drug strategies.
In a 2023 paper, Dr. Krakos’ team used CELLINK’s BIO X to fabricate a multi-material lab-on-chip platform. This is the first use of bioprinting technology for creating microfluidic chips to use with the H69AR cell line. As the team saw the potential of hydrogels adapted to cell line’s drug-resistant phenotype, they experimented with six different compositions of hydrogel inks with different proportions of sodium alginate, agar, chitosan, gelatin, and methylcellulose. The team printed the hydrogels using a three-layer “sandwich”-type lab-on-chip.
The printed lab-on-chip (or cancer-on-chip) model successfully mimicked the lung carcinoma microenvironment characteristics, and all hydrogel compositions promoted prolonged cell survival. The best cell viability and longevity was observed in a 3% sodium alginate + 7% gelatin + 90% NaCl hydrogel composition.
Overall, this shows a lot of promise for improved lung cancer therapeutics, and we look forward to following the researchers’ future studies.
Patch-based therapies for lung regeneration
Biocompatible bi-layered hydrogel patches tailored for dynamic organ mechanisms
Over the past two decades, there has been a persistent endeavor to create therapeutic patches for enhancing the repair and regeneration of human organs. One of the primary hurdles in the development of successful patch-based therapies lies in adapting the patch to the intricate mechanical dynamics of organs. Patch-based therapies have a lot of potential in treating, for example, secondary spontaneous pneumothorax (SSP), which can arise from a multitude of diseases such as: COPD, asthma and tuberculosis. This comes from the ability for the patches to regenerate tissue and fill holes, as seen in the study we highlight below.
Dr. Parth Chansoria and her team at the University of North Carolina have created a patch that features anisotropic and auxetic properties, encompassing the wide range of lung dynamics. The team used the LUMEN X to print a bi-layered patch, with a cell-adhesive GelMA bottom layer and a non-adhesive PEGDA top layer. CELLINK’s technology gave the team the ability to tailor the patches to conform to the mechanics of different organ tissue, particularly lung tissue.
These DLP bioprinted patches were found to be effective in sealing holes and preventing pulmonary air leakage from rat models. The success of these hole-filling patches proves the viability of the design approach used, and its adaptability to meet different organ requirements. All in all, the team’s work shows the potential of patch-based therapies in clinical applications.
Accurate 3D models of lung tumor microenvironments
3D printed lung models for precision drug screening in lung tumors
Drug resistance is one of the greatest challenges in the field of oncology, and the main barrier for successful cancer treatment. Traditionally, anti-cancer drugs are evaluated using 2D cell culture platforms. However, 2D cell culture fails to mimic the complexity of the tumor microenvironment and fall short in predicting drug resistance effectively. Since 90% of chemotherapy failures are related to drug resistance, printing an accurate 3D lung model could help in predicting drug resistance and enable greater success.
In a study by Gebeyehu, Surapaneni, Huang, et. al, a combination of the BIO X and INKREDIBLE was used to formulate bioinks optimized to be printed into scaffolds for drug screening. The team printed hydrogel scaffolds with patient-derived xenograft (PDX) cells to ensure a highly precise recreation of tumoral microenvironments. The study covered cell-cell interactions, nutrient gradients and tissue architecture. The team achieved good printability and shape fidelity while maintaining high cell viability through a series of rheological assessments.
These accurate, bioprinted 3D models represent a leap in the understanding of drug resistance within lung carcinoma. The researchers suggest that by creating in-vitro tumor microenvironments with their hydrogel, they allow for the high throughput screening of various anti-cancer drugs. This leads to both better cancer treatments and allowing researchers to find promising cancer drug candidates faster than before.
Printing lung tissue is advancing our understanding of lung disease
The papers we’ve highlighted above exemplify the power of 3D bioprinting in advancing humanity’s understanding of lung disease and tissue regeneration.
With researchers around the world working on improving lung therapeutics, bioprinting is becoming an increasingly common tool at their disposal, due to its flexibility and ability to mimic human lung tissue. We look forward to continuing following how researchers utilize bioprinting in innovative ways to drive the development of more effective therapeutic approaches.
If you are eager to explore how bioprinting can elevate your own research, book a consultation with our applications scientists.




