2D vs 3D Cell Culture
Two-dimensional (2D) cell cultures grow in flat monolayers on rigid surfaces, while three-dimensional (3D) cell cultures allow cells to organize and interact in 3D environments, more accurately mimicking real tissue environments. This distinction is crucial for research and drug discovery, as 3D cultures better capture the complexity and behavior of living tissues, leading to more predictive and reliable experimental outcomes. Bioprinting further enhances 3D cell culture, enabling the precise and reproducible construction of complex, tissue-like structures
What is 2D Cell Culture?
Adherent 2D cell culture is a traditional laboratory technique where cells are grown as a flat monolayer on a rigid surface, spreading out in only two dimensions. This method dates back over a century and has been the cornerstone of biomedical research due to its widespread adoption because of its simplicity, reproducibility, and low cost.
Advantages of 2D Cell Culture
Straightforward and cost-effective
2D cell culture is simple to establish and maintain, making it accessible for most laboratories, and is therefore a standard approach for routine studies in cell biology, pharmacology, and molecular research.
Fast growing and easy to manipulate
Limitations of 2D Cell Culture
Flat growth environment
Cells in 2D grow on a single rigid plane that fails to mimic the spatial organization and microenvironment of native tissues, leading to altered cell morphology, gene expression, and signaling pathways.
Poor mimicry of in vivo conditions
What is 3D Cell Culture?
3D cell culture allows cells to grow and interact in all spatial directions within a supportive matrix or scaffold, closely replicating the cellular behavior and microenvironment found in living tissues.
Common 3D cell culture models include tissue engineering models, organoids, spheroid cultures, and organ-on-a-chip models. With more accurate modeling of in vivo conditions, 3D cell culture enables improved biological relevance for disease modeling, drug screening, and regenerative medicine research
Advantages of 3D Cell Culture
Mimics tissue architecture
Cell-cell and cell-ECM interactions
Limitations of 3D Cell Culture
Complexity limiting flexibility
Some 3D systems require proprietary materials and lack control in the spatial arrangement of cells, leading to increased costs.
Our 3D bioprinters overcome this limitation by having a user-friendly, open material platform with multiple printheads. This means users can use any material of their own choice, while staying in control of spatial cell arrangement.
Scalability challenges
Some 3D models are difficult to standardize or scale for high-throughput applications.
Bioprinting addresses this by enabling precise, protocol-driven deposition of cells and biomaterials, ensuring consistent structures, from droplets and lattice scaffolds to complex tissues. With controlled volumes, low print-to-print variation, and compatibility with multi-well formats, it supports standardized workflows suited for higher throughput applications.
Impact on research outcomes
The shift from 2D to 3D cell culture has significantly enhanced the physiological relevance and predictive accuracy of experimental outcomes. Compared to 2D systems, 3D models better replicate cellular responses to drugs, disease progression, and therapeutic efficacy. Bioprinting further enhances these advantages by improving scalability, reproducibility, and control over tissue architecture.
| Feature | 2D cell culture | Spheroids | 3D tissue models |
|---|---|---|---|
| Structure | Flat, monolayer of cells | Spherical aggregates | Tissue-like architecture |
| Physiological relevance | Limited; lacks 3D architecture and ECM signaling | Improved; captures basic nutrient and oxygen gradients | High; reproduces tissue-specific microenvironments, stiffness, and signaling dynamics |
| Predictive accuracy | Poor; limited translational relevance | High; improved modeling of in vivo gradients | Excellent; closely replicates native tissue structure and function |
| Drug resistance modeling | Poor; lacks physiological diffusion barriers | Moderate; partial replication of in vivo drug gradients | Excellent; mimics cell-cell and ECM interactions influencing drug penetration and metabolism |
| Scalability | High; compatible with automation and screening platforms | High; suitable for medium-throughput assays | Moderate; requires specialized bioprinting systems but offers enhanced reproducibility |
Drug response differences
Cells cultured in 3D environments have significantly different pharmacological responses compared to cells grown in 2D monolayers. Increased cell-cell and cell-ECM interactions in 3D models contribute to limited drug penetration, altered receptor activity, and enhanced survival signaling, influencing drug sensitivity. Recent studies have shown that cancer cells grown in 3D require higher drug concentrations to achieve comparable cytotoxic effects seen in 2D, offering a better understanding of in vivo molecular changes in response to different treatments.
Disease modeling accuracy
3D cell cultures provide a more accurate representation for studying disease mechanisms by capturing the spatial, biochemical, and mechanical complexity of tissues. 3D cell culture models, including organoids, spheroids, and bioprinted tissues, have been used to understand the roles of cancer-associated fibroblasts in breast cancer, mimic microenvironmental heterogeneity in endometriosis, and model hepatic disease microenvironments.
The gif shows breast cancer cell invasion in TeloCol-10, as described in our Application Note:
Automated Spheroid Invasion Assay Using Collagen from Advanced BioMatrix.

The role of bioprinting in the shift from 2D to 3D cell culture
Enabling reproducible 3D models
Bioprinting enables reproducible 3D models by depositing cells and biomaterials precisely in defined geometries, generating uniform tissue constructs with minimal experimental variation.
Researchers at Ronawk used CELLINK’s BIO X™ and LUMEN X™ to bioprint modular T-block scaffolds that support continuous adult stem cell growth without passaging. This high-throughput, reproducible design minimizes variability and ensures consistent outcomes across experiments.
Scalable solutions for drug discovery and disease modeling
Bioprinting enables scalable, high-throughput fabrication of 3D tissue models that can be used for drug screening and toxicity testing, which are difficult to standardize using manual or spheroid-based methods.
CELLINK researchers utilized the BIO X™ to bioprint mini-liver models composed of HepG2, HUVEC, and LX2 cells embedded in collagen for drug-induced liver injury screening. These models were compatible with automated handling systems, demonstrating scalability and throughput for pharmaceutical workflows.
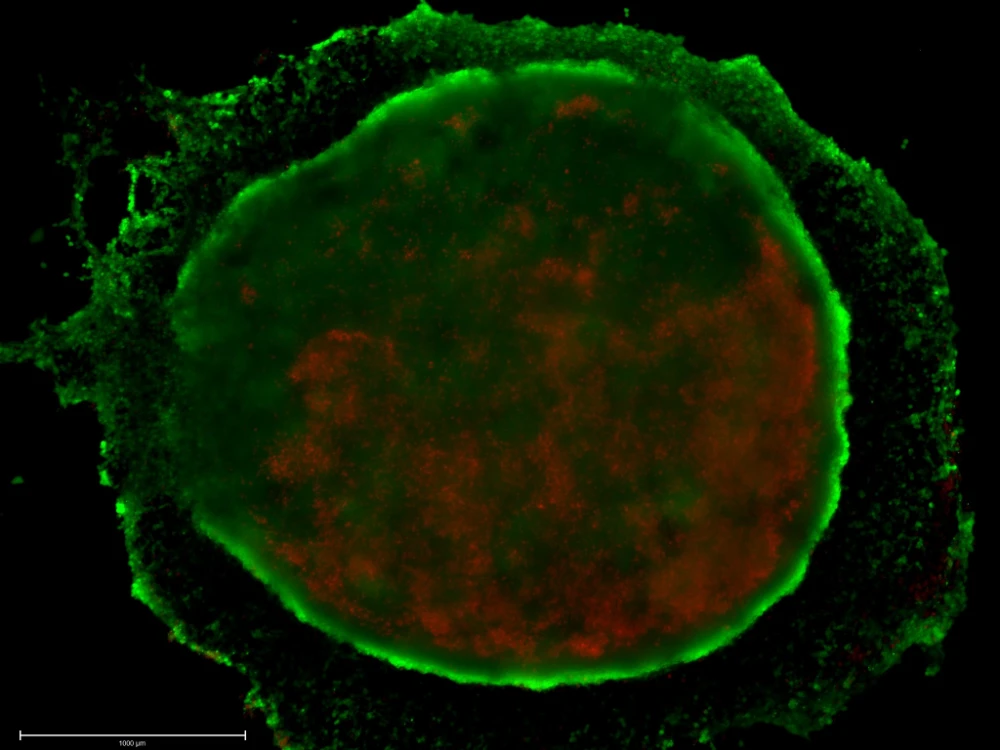
Advancing personalized medicine
Bioprinting allows the fabrication of patient-specific tissues and implants that replicate individual patient anatomy and physiology, paving the way for personalized therapies.
At the University of Gothenburg, researchers used the BIO X™ by CELLINK to print cartilage constructs from patient-derived iPSCs using a nanocellulose/alginate bioink. The resulting tissues expressed key cartilage markers and were structurally indistinguishable from native cartilage, advancing personalized treatment for knee injuries.

Conclusion
While 2D cell culture systems remain valuable for routine studies, 3D cell culture, especially when enabled by bioprinting, delivers greater physiological relevance and predictive accuracy. CELLINK’s bioprinters empower researchers to overcome the limitations of conventional culture systems. With advanced precision, automation, and reproducibility, these platforms are enabling the creation of accurate and personalized models for drug discovery, disease modeling, and regenerative medicine.
Contact an expert to discover how CELLINK’s next-generation 3D bioprinting solutions can enhance reproducibility, scalability, and translational relevance in your research.
References
Kapałczyńska M, Kolenda T, Przybyła W, et al. 2D and 3D cell cultures – a comparison of different types of cancer cell cultures. Arch Med Sci.2018;14(4):910-919. doi:10.5114/aoms.2016.63743
Fang Y, Eglen RM. Three-Dimensional Cell Cultures in Drug Discovery and Development. SLAS Discov. 2017;22(5):456-472. doi:10.1177/1087057117696795
Jensen C, Teng Y. Is It Time to Start Transitioning From 2D to 3D Cell Culture? Front Mol Biosci. 2020;7:33. doi:10.3389/fmolb.2020.00033
Wang L, Hu D, Xu J, et al. Complex in vitro model: A transformative model in drug development and precision medicine. Clin Transl Sci. 2024;17(2):e13695. doi:10.1111/cts.13695
Park CH, Park JH, Suh YJ. Perspective of 3D culture in medicine: transforming disease research and therapeutic applications. Front Bioeng Biotechnol. 2024;12:1491669. doi:10.3389/fbioe.2024.1491669
Lindner N, Blaeser A. Scalable Biofabrication: A Perspective on the Current State and Future Potentials of Process Automation in 3D-Bioprinting Applications. Front Bioeng Biotechnol. 2022;10:855042. doi:10.3389/fbioe.2022.855042
Alwahsh M, Al-Doridee A, Jasim S, et al. Cytotoxic and molecular differences of anticancer agents on 2D and 3D cell culture. Mol Biol Rep. 2024;51(1):721. doi:10.1007/s11033-024-09669-1
Bloise N, Giannaccari M, Guagliano G, et al. Growing Role of 3D In Vitro Cell Cultures in the Study of Cellular and Molecular Mechanisms: Short Focus on Breast Cancer, Endometriosis, Liver and Infectious Diseases. Cells. 2024;13(12):1054. doi:10.3390/cells13121054
Shuford S, Lipinski L, Abad A, et al. Prospective prediction of clinical drug response in high-grade gliomas using an ex vivo 3D cell culture assay. Neuro-Oncol Adv. 2021;3(1):vdab065. doi:10.1093/noajnl/vdab065




