Bone tissue
Tuning the mechanical microenvironment for bone formation
Bone tissue is an inorganic-organic nanocomposite composed of hydroxyapatite nanocrystals embedded within collagen type I fibrils. The bioinks we offer for bone tissue engineering, CELLINK BONE and GelXA BONE, are designed to resemble the chemical composition of bone. Our bioinks enable bone cells to incorporate during the bioprinting process and their homogeneous distribution throughout the scaffold provides the porosity characteristic of bone tissue.
Products used:
Aim
The aim of this study is to demonstrate the mechanical versatility of tissue constructs bioprinted with GelXA BONE bioink, and to show the correlation between mechanical properties of cell microenvironment and cell behavior.
Rheology
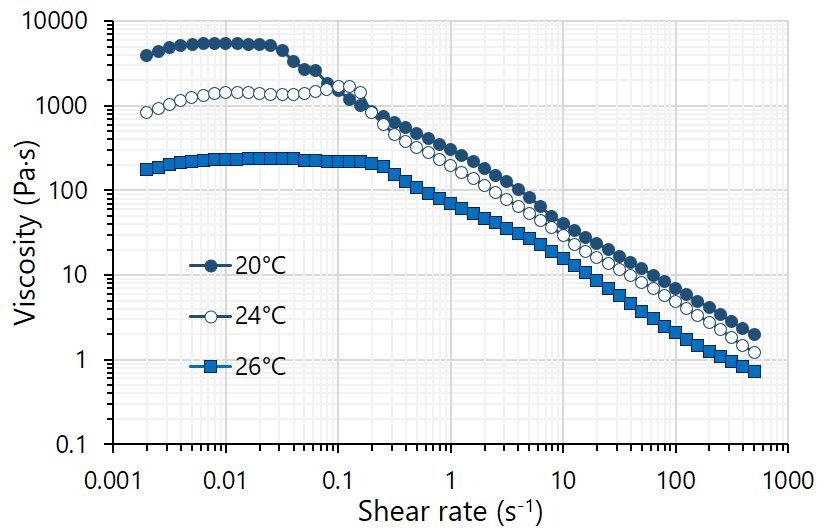
GelXA bioinks are gelatin-based hydrogels which are designed to be less sensitive to the thermal environment during printing. They are shear thinning in ambient conditions and enable smooth printing at temperatures between 20 and 26°C with minor adjustments to printing pressure, as shown in the figure beside for GelXA BONE. Because GelXA bioinks contain xanthan gum as a thickener, any unfavourable flow after printing is substantially reduced, producing constructs with higher resolution and well-defined shapes.
Mechanical properties
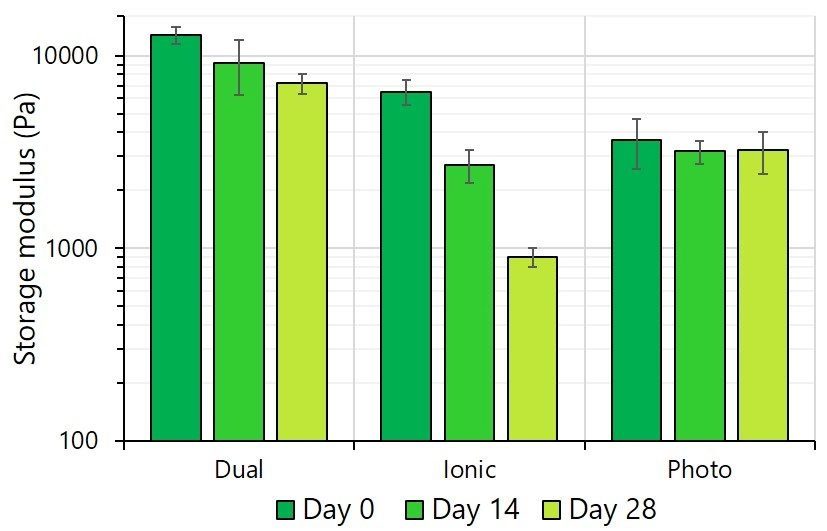
The stiffness of GelXA BONE constructs, expressed as storage modulus, is dependent on the crosslinking method used. As expected, dual crosslinking provides the highest initial stiffness, while ionic crosslinking results in medium stiffness and photocrosslinking results in the lowest stiffness. Stiffness decreased over the 28-day experiment in all cases: the drop was negligible for the photocrosslinked bioinks, moderate for dually crosslinked and very steep for ioinically crosslinked. The hypothesis is that ionic crosslinking is not stable over time due to Ca2+ ion diffusion outside the constructs.
Osteoblast network formation
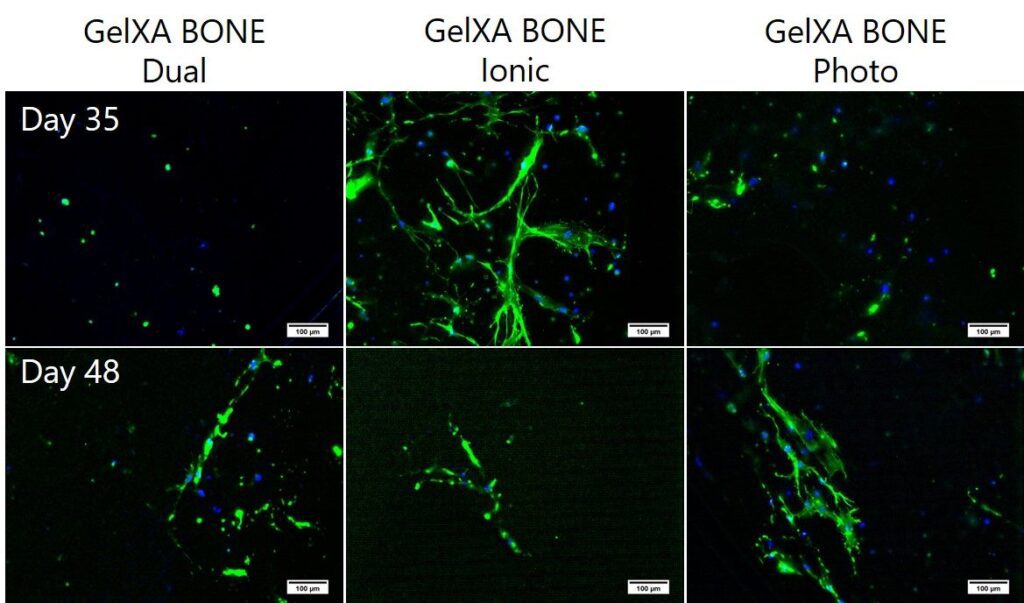
Osteoblasts in GelXA BONE constructs, bioprinted with the BIO X, demonstrate physiological-like behavior (stretched cells forming a network), but you can observe clear variance for differently crosslinked samples. The softest ionically crosslinked constructs offer the best microenvironment for cell spreading, and extensive network formation is seen already at Day 35. Cell spreading is less pronounced in stiffer photocrosslinked and dually crosslinked constructs, and network formation is observed later on at Day 48.
Expression of osteogenesis markers
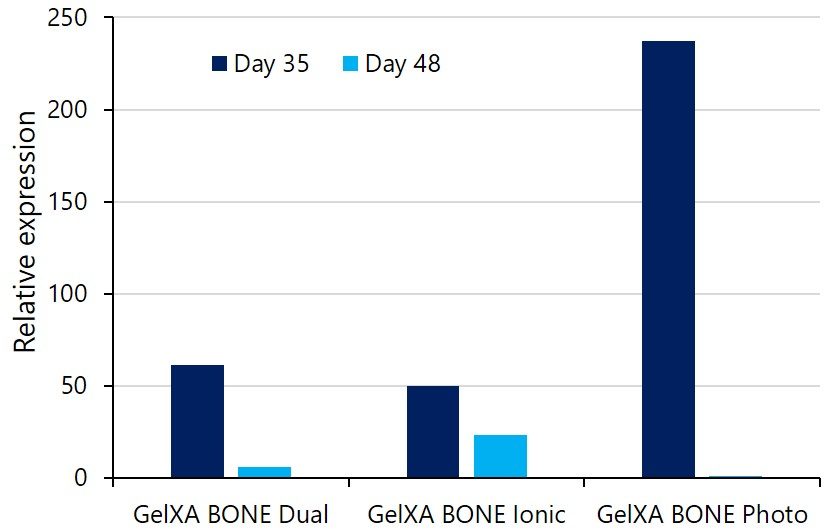
Osteoblasts express a matrix material called osteoid, which is mostly composed of collagen type 1. Collagen type I secretion is an early marker of osteogenesis. Osteoblasts in all GelXA BONE constrcuts show collagen type I expression at Day 35, corresponding to 21 days after the addition of differentiation media (see Figure beside). Collagen type I expression later decreases up to day 48. Interestingly, we observe the highest expression in osteobalsts embedded into the most mechanically stable constructs, in this case, photocrosslinked ones.
Conclusion
This study demonstrates that GelXA BONE offers great versatility, enabling a wide range of bioprinting temperatures and fine mechanical tuning of the bone cell microenvironment through different crosslinking techniques. We successfully cultured osteoblasts in bioprinted constructs for 48 days and found early markers of bone formation.




