薬物性肝障害のコラーゲンモデル
組織構造、細胞間相互作用、細胞再編成を担うコラーゲンは、組織工学研究で広く使用されています。その豊富な結合部位、容易なゲル化、魅力的な分解特性により、肝臓を含むほとんどの組織モデルに対応する汎用的なバイオインクです。
最近の研究では、CELLINKの技術チームが、急性肝不全、肝炎、線維症の主な原因である薬物性肝障害(DILI)を評価するために、コラーゲン「ミニ肝臓」のバイオプリントを行いました1。I型コラーゲンは、肝組織に多く存在し、3次元細胞培養に適した特性を持つことから、この研究に選ばれました3。さらに、コラーゲンは、均一に架橋し、液滴でプリントすることができるため、他のバイオマテリアルとは比べものにならないほど優れています。ミニ肝臓モデルには、HepG2、LX2、HUVECなど複数の肝細胞と非肝細胞を用い、アルブミン分泌、脂質蓄積、肝障害のマーカーとして知られるアラニンアミノトランスフェラーゼ活性などの肝機能を模倣するよう作製されました。各モデルは、細胞を含んだコラーゲンI液滴の中に、別の細胞を含んだコラーゲンI液滴を入れ構成されました。液滴のカプセル化が採用されたのは、適切な栄養素と酸素の勾配を備えた高機能の肝臓モデルを作製するためです。カプセル化はまた、細胞のコミュニケーション、細胞配列の制御、組織の層間の異なる剛性を研究する機会にもなりました。
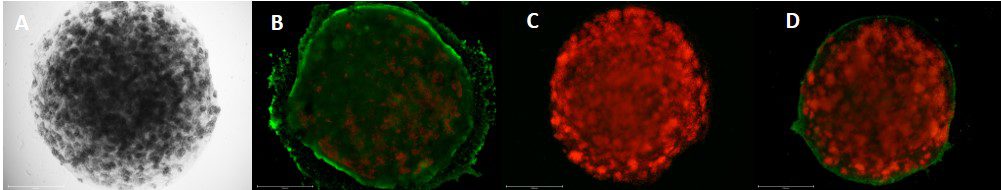
肝毒性評価では、DILIに関連する薬物であるアセトアミノフェンやフルタミドの高用量に対して、ミニ肝臓モデルが有害な反応を示すことがわかりました。これらの結果は、ミニ肝臓モデルが、薬物毒性の正確な予測因子として、また動物実験、2次元細胞培養、Organ-on-a-chipモデルに代わるハイスループットな方法として、薬物スクリーニングに実装できることを示唆しています。
コラーゲンの多様性は、他の多くの研究においてバイオインクを選択する要因となっています。例えば、コラーゲンの分解パターンやレオロジー(流体力学的)特性は、肝臓の損傷や炎症を研究する場合、重要になる可能性があります。後者は肝臓の圧痛や肥大と関連しており、線維症の異なるステージをモデル化し、さまざまな硬さの組織における細胞移動を評価する将来の研究の保証となり得ます。
コラーゲンを使用したバイオプリンティング
コラーゲンは、その汎用性にもかかわらず、温度、圧力、レオロジーの変化に敏感な、難しい特性のバイオマテリアルであることはよく知られています。しかし、コラーゲンのプリント適合性を高める方法はいくつかありますので、ご安心ください。ここでは、その方法をご紹介します:
- 低粘度コラーゲンを増粘剤またはアルギン酸などの他のバイオマテリアル(生体材料)と組み合わせる
- 低粘度コラーゲンをFRESH LifeSupport や同様の微粒子分散液中でバイオプリンティングする
- 機械的剛性とプリント適合性を高めるために、光架橋性コラーゲンであるColMAを検討する
- 温度制御プリントヘッド を使用して、コラーゲンをバイオプリンティングまたはドロップレットプリンティングする
体内で最も重要なタンパク質の1つであるコラーゲンは、組織工学の研究において重要な役割を果たしています。ヒトの心臓の一部の再構築2、細胞傷害性T細胞による腫瘍の標的化4、アンチエイジング機構の発見など、研究への応用の可能性は数多くあります。さて、あなたはコラーゲンを使って何をプリントしますか?
参考文献
- David, S., and Hamilton, J. P. Drug-induced Liver Injury. S. Gastroenterology & Hepatology Review. 2010; 6: 73–80.
- Lee, A., Hudson, A.R., Shiwarski, D.J., et al. 3D bioprinting of collagen to rebuild components of the human heart. 2019; 365(6452): 482–487.
- Martinez-Hernandez, A., and Amenta, P. S. The hepatic extracellular matrix. I. Components and distribution in normal liver. Virchows Archiv. A pathological anatomy and histopathology. 1993; 423(1): 1–11.
- Pruitt, H. C., Lewis, D., Ciccaglione, M., et al. Collagen fiber structure guides 3D motility of cytotoxic T lymphocytes. Matrix Biology: Journal of the International Society for Matrix Biology. 2020; 85-86: 147–159.