がん研究へのバイオプリンティング活用事例
がんは、細胞の異常な増殖を特徴とする病気です。世界で2番目に多い死因であり、2018年には推定960万人、つまり6人に1人ががんで死亡しています。
がんは体のどこからでも発生し、他の多くの部位に転移する可能性があります。がんでは、健康な細胞がDNAの変化を受け、制御不能な細胞分裂を始めます。その結果、細胞が過剰に増殖し、その部位に必要な機能が阻害され、腫瘍が発生します。腫瘍ががん性である場合、悪性腫瘍と定義され、隣接する組織に侵入し、体全体に広がり続け、身体機能に混乱をもたらす能力を持っています。また、がんは、殺傷シグナルを無視して私たち自身の免疫システムを回避し、周辺の他の細胞に影響を与え、酸素供給と老廃物除去のネットワークを形成することによって、より多くのがん細胞の増殖を促進するという不思議な能力を持っています。
多くの人が苦しみ続ける中、様々ながんが、医師たちの診断と治療の課題として残り続けています。この10年間で、がんの治療と診断ツールの開発には大きな進歩がありましたが、特に有効な抗がん剤の開発においては、まだまだ改善すべき点があります。従来、製薬業界の研究者は、抗がん剤の試験に2次元細胞培養を利用してきました。2Dモデルは、これまでの理解の多くを導き、現在に至るまで重要な役割を果たしてきましたが、がん性組織で発生する細胞間相互作用を正確に再現することはできません。3Dバイオプリンティングは、平面的な細胞培養から、生理学的に適切なin vitroモデルの開発を進める上で、非常に有望で可能性を秘めた手法です。具体的には、自動化、精密化、バイオマテリアルの巧みな利用により、バイオプリンティングは、複雑な腫瘍の微小環境を再現性よく、高スループットに模倣する方法を提供します。
当社の吉江は、先日のウェビナーで、がん研究の世界で3Dバイオプリンティングを探求する際に、研究者が考慮しなければならない問題について議論しました。使用するバイオインク、細胞種、構造設計からプリント条件といった全てのものが、生化学的相互作用や、細胞間の相互作用、そしてモデル全体の精度にも影響を与える可能性があるのです。また、がんの微小環境、がんの転移、がんの血管新生、薬物スクリーニングなど、現在バイオプリンティングで研究されている具体的な分野についてもご紹介しています。
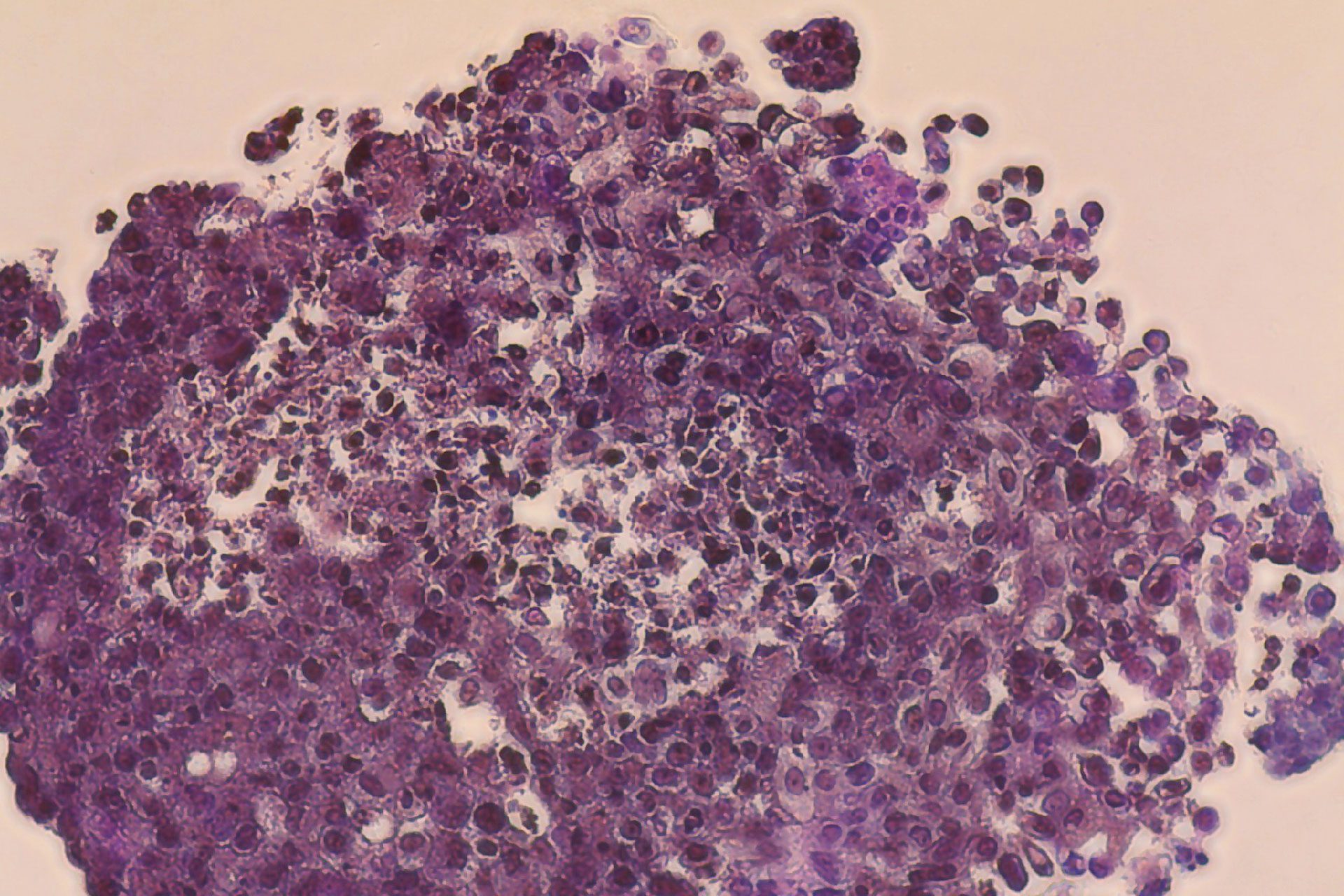
ウェビナーでは、現在CELLINKのお客様が取り組んでいる、非小細胞肺がんモデルのスフェロイドに関する興味深いプロジェクトについても説明しています。研究チームは、患者由来の腫瘍細胞とがん関連線維芽細胞(CAF)細胞をバイオインクに共培養し、INKREDIBLEを使用してプリントしました。彼らは、プリント適合性、細胞生存率、細胞間クロストーク、機械的特性を調べ、スフェロイド形成が時間とともに増加し、より生理学的に近いがんモデルであるin vitro腫瘍共培養スフェロイドが生成されることを確認しました。
これに加えて、CELLINKの研究チームが、先日、同系腫瘍モデルを開発するための実験を実施しました。彼らは、BIO Xを使用してマウス肺がんモデルを開発し、T細胞の殺傷効率を評価しました。3DバイオプリントされたがんモデルでT細胞の細胞傷害性アッセイを行うことで、チェックポイント阻害剤をより効率的かつトランスレーショナルにスクリーニングできることを実証したのです。今回の研究についての詳細や、吉江によるウェビナーは下記リンクよりご覧いただけます。