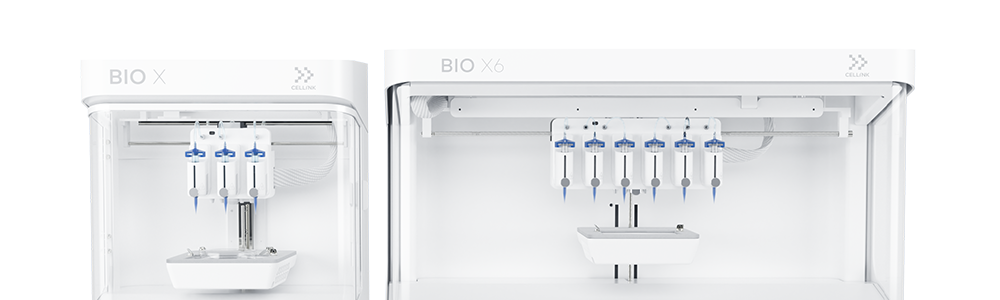
Leading the way in 3D bioprinting
We Create the Future of Health
Unlock greater insights by leveraging the power of 3D cell culture through award-winning 3D bioprinters and carefully sourced biomaterials and bioinks.
Areas of Impact
The 3D bioprinting revolution is disrupting research across critical research areas and within the bioprinting industry.
3D Bioprinting, Explained
3D bioprinting is an additive manufacturing process similar to 3D printing – it uses a digital file as a blueprint to print an object layer by layer. But unlike 3D printing, bioprinters print with cells and biomaterials, creating organ-like structures that let living cells multiply. Although bioprinting is a relatively new technology, it has huge potential to benefit industries like regenerative and personalized medicine, drug discovery and cosmetics. Find out how 3D bioprinting works here.
Trusted by
Industry Leaders
Our products are used in a number of industries and sectors by leaders in their respective fields all around the world.